Read on to explore the intricate process behind Volpi’s development of a cutting-edge qPCR module. Our project team reveals the unique challenges they faced and the innovative solutions they employed, showcasing Volpi’s strengths in delivering tailored, high-quality measurement systems for our clients.
Project Team: Karin Ettlin – Project Manager; Peter Dietiker – Senior Test Engineer; Michael Brunotte – Senior Systems Engineer.
Can you describe the project and its goals?
Karin: Our customer requested a cost-effective alternative to their existing qPCR device without compromising on performance or reliability. They sought a partner with proven expertise in optics-based PCR measurement systems that could engineer a cost-effective solution without compromising on the stringent performance benchmarks essential to their application. They were also looking for a flexible partner who keeps an open mind and approaches problem-solving unconventionally. We rose to the challenge, aiming to reach a high level of accuracy at a challenging cost target.
How did you proceed to address customer requirements?
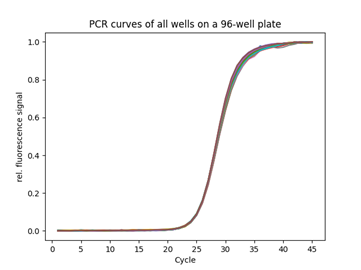
Michael: At first glance, when we reviewed the customer assay and commercial requirements, we immediately knew this would be challenging but could be done. First, our team of solution architects and PCR experts worked together to break down the complexity and derive requirements like “high Cq uniformity” into requirements on the precision of thermal cycling, fluorescence readout and Cq calculation algorithm. We then kicked off brainstorming theoretical possibilities to resolve the challenge. The team got fired up by the difficulty of the requirements, so this positive attitude started to bear fruit when the contours of theoretical solutions came to light. The next big challenge was how to bring theory into reality. We knew the project’s success hinged on our ability to integrate precise thermal control with high-fidelity optical readouts. By collaborating closely with our suppliers and leveraging our expertise in system integration, we engineered a thermo-cycler module with adequate temperature zones and real-time control, ensuring sufficient temperature uniformity across the PCR plate. One problem solved.
Peter: On the optics side, we selected a scanning solution for its superior inherent uniformity since we had enough time to acquire data. We had to ensure that the scanning head was moving precisely and repeatedly over the PCR plate and was insensitive to changes in ambient temperature to reduce noise and drifts in the optical measurement. Once this tough mechanical issue was resolved, we combined and synchronized the optics with the thermal cycler.
How was the team able to demonstrate the desired CQ uniformity to the customer?
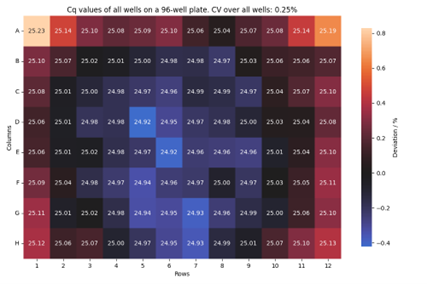
Karin: We can do this by running customers’ assays on our prototypes and directly comparing the results with those on their current systems. For that, we transferred their assay to our testing lab. We also set up reliability tests, where we ran thousands of PCRs and showed that the performances were consistently maintained over time. We aim to ensure our modules work well with customers’ assay portfolios to produce reliable results.
Peter: It’s a constant optimization process until you reach the desired outcome. It is really a trial-and-error approach. For that, I ran many PCR to test and tune our system and verify that our module was operating as expected. I also had to adapt the PCR parameters, such as temperatures, ramps, and time, to ensure the PCR amplification was effective. At this precision level, any small pipetting or plate sealing errors immediately appear in the results. So, you need a precise instrument and skilled lab technicians who can prepare PCR plates with minimal well-to-well variability.
Michael: Making all constituent parts – optics, mechanics, thermal engineering, application testing, and data processing – work in sync, consistently and reliably to produce the desired outcome was key.
How did the team work to address customers’ needs and challenges?
Karin: We had weekly technical meetings to discuss our progress and share customers’ feedback. The project was very fluid, and targets were constantly moving as demands and requirements changed throughout it. We addressed them flexibly, focusing solely on customer satisfaction. So, frequent touchpoints and open communication helped us better understand the customers’ needs.
Peter: The customer really appreciated our flexibility. After all, it was one of the reasons they chose us. I would also add another aspect that made the collaboration successful: besides open and frank information sharing – the customer challenged us, and we challenged the customer. This mutual challenge, tug-and-pull, was very positive and constructive as we re-evaluated assumptions and learned from each other. This type of collaboration led to a better product), customer satisfaction, and trust.
What techniques and tools were crucial in this complex project?
Karin: We used agile methodology. That meant we had a fixed team dedicated solely to this project. This was greatly beneficial for team chemistry and communication. We acutely noticed the benefits during the testing phase due to complexities and resource limitations. What I mean is that we had to evaluate the module with an assay to ensure effective pairing. However, assay testing is expensive, and few assays were available for many tests. The agile method assumes that we sit together, discuss, and prioritize which tests are essential to run and which are “nice to have.”
Michael: Indeed, that was beneficial. Another aspect of this methodology that worked well is virtual management. We had a dedicated project space where project timelines and milestones were visualized. The issue and task management board was also very helpful. Even simple things, such as Post-it notes to write down the challenges we encountered and assign tasks with timelines, were highly beneficial.
Karin: Of course, we use professional, software-based project management tools. However, the Agile approach ensured we operated in concert, leaving nothing to chance.
Can you share a moment in which the team was collectively proud?
Peter: When we got the first results from testing that matched the requirements, it was truly a moment that we felt proud of. We still knew a long road lay ahead to achieving the reliability level, but we understood something clearly: at last, there is a light at the end of the tunnel. We were a bit nervous when we visited the customer with a lab prototype for testing and technical discussions. Nevertheless, the testing went very smoothly, and when we saw the customer’s reaction, that was another proud moment for us as a team.
Karin: Indeed, I remember that very well. The same thing happened when we visited the customer for the second time with Alpha Prototype. The testing went extremely smoothly, the module worked perfectly, and we had plenty of free time to engage in productive activities with the customer. The customer was pleased, and we gained a lot of trust from them, which was also very rewarding. We have combined different capabilities into a single, orchestrated result for the customer. This was very rewarding, and we look forward to collaborating with the customers in the next product development phase.

